|
Chemguide: Support for CIE A level Chemistry Learning outcome 30: Hydrocarbons 30.1: Arenes Learning outcome 30.1.4 This statement is about the position of substitution in a benzene ring when there is already something else attached to the ring. Before you go on, you should find and read the statement in your copy of the syllabus. Read the page about electrophilic substitution into already substituted benzene rings. You will have to learn the directing effects of the groups mentioned in the syllabus. In fact there is a pattern, and it isn't difficult.
Notice:
You do not have to explain any of this. | ||||||||||||||||||||||
|
Note: The activating or deactivating effects of groups isn't mentioned in the syllabus at this point. However, you will need to know about it when we talk about reactions of particular aromatic compounds later in the syllabus. | ||||||||||||||||||||||
|
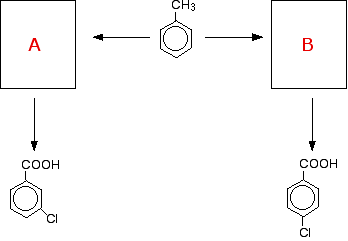
At its simplest, you could be asked to draw the structure for the reaction between, say, nitrobenzene and a heated mixture of concentrated nitric and sulfuric acids. You will have learnt that the nitro group directs into the 3 position, so you would add a second nitro group there. This being CIE, of course, they are unlikely to ask you anything so simple! Here is a more likely question, which is going to need a bit more thought. Suppose you were given this incomplete flow scheme for a series of reactions:
You will have learnt that a methyl group directs into the 4-position, and that a COOH group directs into the 3-position. The problem is to work out what intermediate compounds have to go into the boxes marked A and B. You would also almost certainly be asked for the conditions for the various reactions involved as well. If you look at what you have to do, you need to convert the methyl group into a COOH group, and substitute a chlorine atom into the ring in two different positions. So what reactions do you need to do?
The question is then about which order you do these reactions in.
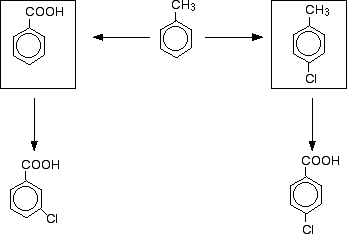
That means that to get the left-hand product, you would have to oxidise the methyl group first, and do the substitution reaction afterwards. To get the right-hand product, you would have to do the substitution first, and then oxidise the methyl group afterwards. The flow scheme would then look like this:
Provided you know about the directing effects of the groups already on the ring, and provided you know the conditions for these reactions, there is nothing difficult about a question like this. Don't be put off by CIE's tendency to dress a fairly straightforward question up to make it look more difficult than it is. Some other things you might come across in questions of this sort Electron donating and withdrawing groups You may remember that somewhere in what I have asked you to read, I have talked about the tendency of methyl groups to "push" electrons towards the ring. Another way of saying this is that the methyl group is an electron-donating group. The nitro group is an electron-withdrawing group - it pulls electrons away from the ring. The reasons for these effects are beyond what you need for CIE purposes, but in the past CIE used these terms when telling you about the directing effects. As a general rule:
| ||||||||||||||||||||||
|
Note: There is an exception to this generalisation. Halogens are electron-withdrawing groups (because of their electronegativity), but they direct into the 2- and 4-positions. Halogens aren't included in CIE's list of groups and so you don't need to know that. | ||||||||||||||||||||||
|
Mechanisms for multiple substitution There is no indication that CIE will ask you for these, but it is possible that they could. The only difficult thing here is in getting the structure of the intermediate ion right. So that this doesn't take you by surprise if it comes up, look again at the link I gave you earlier on this page - electrophilic substitution into already substituted benzene rings. This time, look at the way the mechanisms are written for the substitution of a nitro group into methylbenzene, and into nitrobenzene. In the other examples on that page, look carefully at the exact way the intermediate ions are drawn.
© Jim Clark 2020 |
||||||||||||||||||||||