|
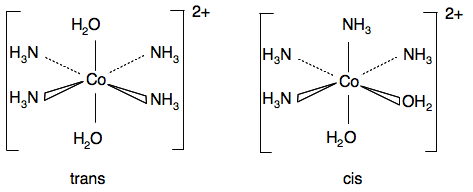
Chemguide: Support for CIE A level Chemistry Learning outcome 28: Chemistry of transition elements 28.4: Stereoisomerism in transition element complexes Learning outcomes 28.4.1 and 28.4.2 These statements are about stereoisomerism in transition metal complexes. Read the statements in your copy of the syllabus as you go along. Statement 28.4.1 Geometric isomerism You will find the square planar example from the syllabus in a short paragraph about half way down the page about shapes of complexes. Don't read any more of that page for the moment. The other more complicated octahedral cases aren't mentioned on that page. For [Co(NH3)4(H2O)2]2+, there are two possible arrangements of the ligands. In one the two water molecules are on opposite sides of the complex, and in the other they are at 90° to each other.
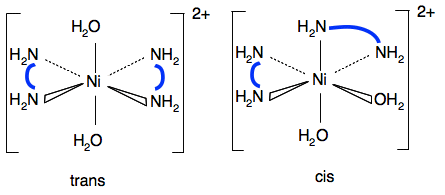
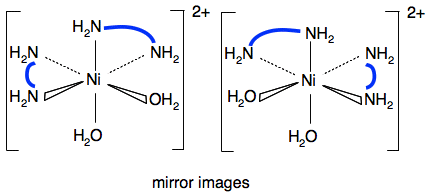
The other example of geometric isomerism in an octahedral complex is [Ni(H2NCH2CH2NH2)2(H2O)2]2+. This has two molecules of 1,2-diaminoethane (a bidentate ligand) and two water molecules attached to the nickel ion. I have tried to make the diagrams less cluttered by replacing the CH2CH2 by a thick blue line.
Again, the water molecules can be opposite each other or at 90°. The structure of 1,2-diaminoethane is shown on the introductory page on complex ions which you probably looked at when you were reading the page about shapes. Optical isomerism Now go back to the page about shapes of complexes and read the section about optical isomerism. The two examples quoted in the syllabus are: [Ni(H2NCH2CH2NH2)3]2+
This has optical isomers and is exactly like the general case at the bottom of the page you have just read. There was a question in the specimen paper for this syllabus which expected you do draw these two isomers. They gave you the formula for 1,2-diaminoethane and asked you to draw 3D diagrams for the two structures. Their mark scheme drew the diagrams in three different ways. The two which are closest to how I have done it are:
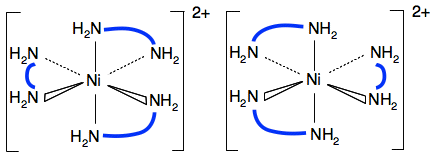
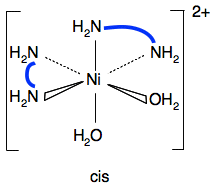
[Ni(H2NCH2CH2NH2)2(H2O)2]2+ One of the geometric isomers also shows optical isomerism - the one with the water molecules at 90°.
If you draw the mirror image of this you get:
Imagine rotating the right-hand one around the vertical axis by 180° so that the 1,2-diaminoethane ligands in the horizontal plane line up. If you do that, the other 1,2-diaminoethane will now be coming out towards you, and the water molecule going back away from you on the right-hand side. There is no way of getting the two diagrams to line up exactly. I find this a real struggle to imagine, and there is a much simpler way of looking at it. Instead, look for a plane of symmetry in the molecule. Is there any plane that you can draw through the molecule where one side of it is an exact mirror image of the other one? In this case, you can't find one. If there is no plane of symmetry then there are optical isomers. The other geometric isomer does have a plane of symmetry.
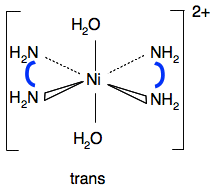
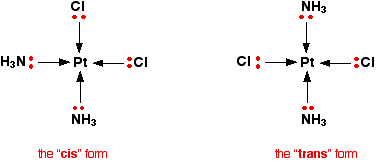
If you imagine a plane drawn horizontally through the nickel and the two bidentate ligands, the top section is an exact mirror image of the bottom. So this one won't have optical isomers. Statement 28.4.2 This statement wants you to think about the polarity of the various molecules we have looked at above. It would be a good idea to read the short section about "Polar bonds and polar molecules" on the page about Electronegativity before you go on. You will find what you want about a third of the way down the page. Let's look again at all the structures drawn above. This diagram for the geometric isomers of Pt(NH3)2Cl2 comes from the Chemguide page you have already looked at.
Chlorine is fractionally more electronegative than nitrogen (Cl 3.16; N 3.04). In the trans form, the effects of the nitrogens and chlorines are cancelled out because the chlorines are opposite each other, as are the nitrogens. In the cis form, that isn't happening. There is a general tendency for the electron density to increase towards the chlorines, and decrease around the hydrogens in the ammonias. (The nitrogen-hydrogen bonds are, of course, polar in themselves.) This leaves the chlorines slightly negative and the hydrogens slightly positive. The cis isomer is polar.
The oxygen in the water molecules is the most electronegative element present, and the hydrogens attached to oxygens will have the greatest degree of positiveness. In the trans form the water molecules are directly opposite each other, and although particular bonds will be polar, the ion overall won't have any polarity. The effects of the two water molecules cancel each other out. The same is true of any polarity due to the ammonia molecules. In the cis form, the two water molecules don't cancel each other out because they are on the same side of the ion. The ammonia molecules are all on the other side. The net polarity of the molecule will be a combination of the effects of the ammonia and water ligands. | |
|
Note: I am not confident about predicting the net effect on the polarity of the ion. There are twice as many ammonia molecules as water molecules, but the polarity of the N-H bond is less than the O-H bond. All I am happy to say is that the ion will show polarity. | |
|
This is exactly the same as the cobalt case above. The important bits are the waters and the nitrogens. The fact that the nitrogens are bridged with CH2CH2 groups is irrelevant. So the trans form won't have any overall polarity, and the cis form will. In the cis forms the oxygens and nitrogens aren't arranged so that they cancel each other out on opposite sides of the ion.
In both isomers, the nitrogens area arranged evenly around the ion. The overall effect will be no polarity.
© Jim Clark 2020 |
|