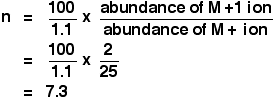|
Chemguide: Support for CIE A level Chemistry Learning outcome 22: Analytical techniques 22.2: Mass spectrometry Learning outcomes 22.2.3, 22.2.4, 22.2.5 and 22.2.6 Statement 22.2.4: Fragmentation patterns Start by reading about fragmentation patterns on this page in the Mass Spectrometry section of Chemguide. You might want to have a quick look back at how a mass spectrometer works, but you don't need to remember any of it. You need to make sure that you could identify which ions might have produced given lines in a simple mass spectrum. And also (and this is more difficult) make reasonable predictions about what lines you might find in the spectrum of a named organic compound. One question asked by CIE wanted you to suggest two small neutral molecules (with a relative molecular mass less than 30) which might be formed during the fragmentation of alcohols. If you try to work this out by imagining what the fragmentation might produce in a logical way, this is really difficult - if not impossible. In fact, rearrangements can happen, so virtually any small neutral molecule containing only carbon, hydrogen or oxygen was allowed. The mark scheme allowed any two from H2, H2O, CO, C2H4, C2H2, CH4. Statement 22.2.3: The M+ peak Now go on to read about molecular ion peaks. There isn't much to read, because you do not need to read the second half of the page about the results from high resolution mass spectrometers using very accurate relative isotopic masses. Statement 22.2.5: The M+1 peak You should read the page about the M+1 peak. This is one of the commonest questions CIE ask about mass spectrometry. They expect you to be able to calculate the number of carbon atoms in a molecule from the ratio between the heights of the M and M+1 peaks. There is a formula for doing this which isn't mentioned on the Chemguide page. You MUST learn this. They commonly ask you to do this at the start of a longer question, perhaps including NMR, and if you can't work out the right number of carbon atoms, you may well not be able to complete the rest of the question. The formula CIE want for the number of carbon atoms, n, is:
For example, if you were told that the relative heights (abundances) of the M+ and M+1 peaks were in the ratio of 25:2, you just have to slot these into the equation - taking care that you get them the right way up!
Questions set using this formula don't seem to produce nice tidy whole numbers. Obviously, there can't be 7.3 carbon atoms per molecule, so you just take the nearest whole number - in this case, there must be 7 carbon atoms. What if you put the numbers in the wrong way up? You will just get a silly answer - in this case, 1136 carbon atoms. If this happens to you, try again with the numbers the other way up. Statement 22.2.6: The presence of chlorine or bromine atoms This is another commonly asked topic in CIE questions. You should read the page about mass spectra - the M+2 peak. The presence of chlorine Notice that the presence of a single chlorine atom causes the M+ and M+2 peaks to have relative heights (abundances) of 3:1, because there is approximately 3 times as much of the Cl-35 isotope as of the Cl-37. Take care to get this ratio the right way round. CIE Examiner's Reports keep saying that some students got the ratio the wrong way around. That is a silly way of wasting marks! You will be expected to know what happens if you have two chlorine atoms. You get M+, M+2 and M+4 peaks in the ratio 9:6:1. Learn that! Despite a comment in an Examiner's Report, if you aren't a mathematician, this isn't easy to work out quickly. Another question told you that a compound contained 4 chlorine atoms, and asked you how many molecular ion peaks there would be. The Examiner's Report commented that few candidates actually got this right. It isn't exactly difficult! You just need to work out how many combinations of the isotopes of chlorine you might have. The molecule might have any of the following combinations of isotopes:
There are five different totals possible. If you add these to the masses of the other atoms in the molecule, you will have five different molecular ion peaks. The presence of bromine Make sure that you recognise that M+ and M+2 peaks of approximately equal heights suggest the presence of a single bromine atom in the molecule. The best way of practising all this (especially in interpreting mass spectra to identify simple molecules) is to go through past papers. The statements in the 2022 syllabus are essentially identical with the previous syllabuses, and so you can look at any question from 2007 onwards. In the past, mass spectrometry involving organic molecules wasn't asked until the second half of the course. You will have to find older questions by looking at versions of paper 4, and probably won't be able to do most of the question. Just pick out the bits about mass spectrometry.
© Jim Clark 2020 |